The formula for assay calculation by titration is based on the principle of chemical equivalence, which states that the number of moles of reactant in a reaction is equal to the number of moles of another reactant.
This technique used to work out the concentration of an unknown solution based on its chemical reaction with a solution of known concentration.
The titration process usually involves adding the known
quantity of a solution (the titrant) to a unknown solution (the analyte) until
the reaction is complete. To calculate the concentration of the analyte, you
measure the volume of titrant used.
Assay by Titrations = [Titrate value of (sample - blank) x M
x F x 100 x 100] / [Ws x (100- LOD)]
Where,
- M = Molarity of Volumetric Solution,
- Ws = Weight of solution,
- F = Factor for drug substance,
- LOD = Loss on Drying.
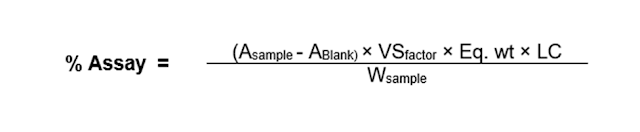
Assay Calculation Formula by Titration Method
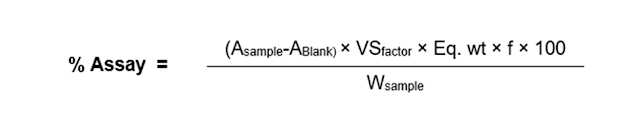
Or
Where,
- A = reading (mL),
- VSfactor = factor of VS (volumetric solution),
- W = weight (mg),
- LC = label claim,
- f = factor of the titrant
- Eq. wt = equivalent weight (molecular weight/ Valency).
Equivalent weight is the mass of the substance in grams that reacts with one mole of the titrant.
% Assay (on anhydrous basis) = (Assay × 100) ÷ (100-LOD/WC)
Where,
- LOD = Loss on drying
- WC = Water content
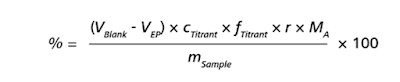
For a back-titration with a blank, use the following formula:
- VBlank: Volume of the equivalence point from the blank determination in mL
- VEP: Volume at the equivalence point in mL
- cTitrant: Nominal titrant concentration in mol/L
- fTitrant: Titer factor of the titrant (unitless)
- r: Stoichiometric ratio (unitless)
- MA: Molecular weight of analyte A in g/mol
- mSample: Weight of sample in mg
- 100: Conversion factor, to obtain the result in %

Post a Comment